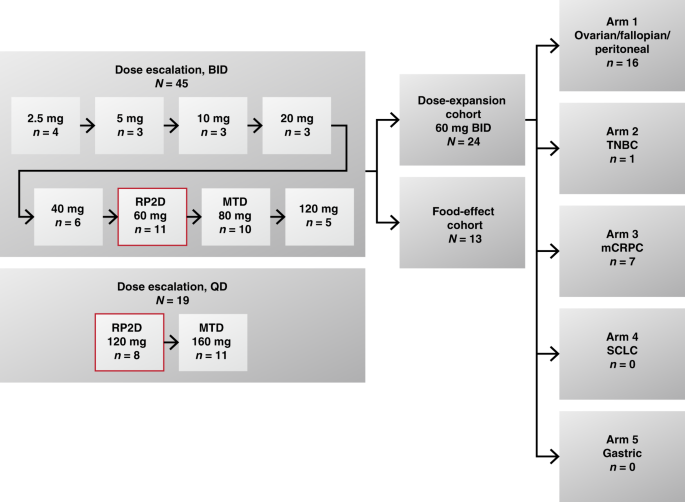
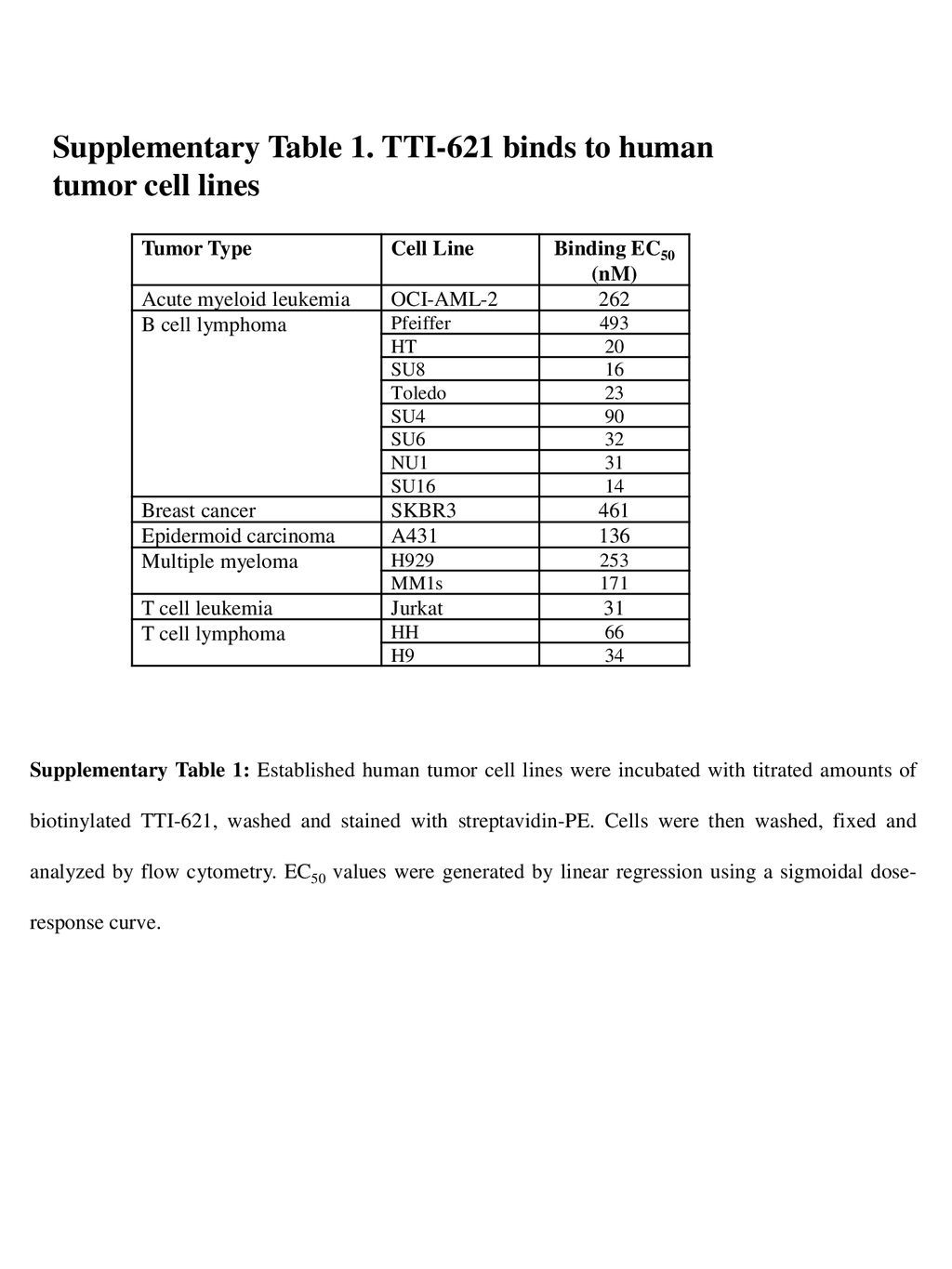
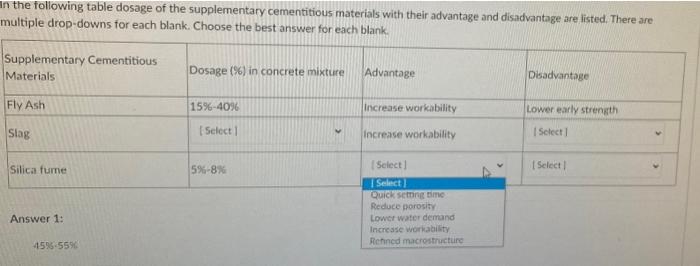
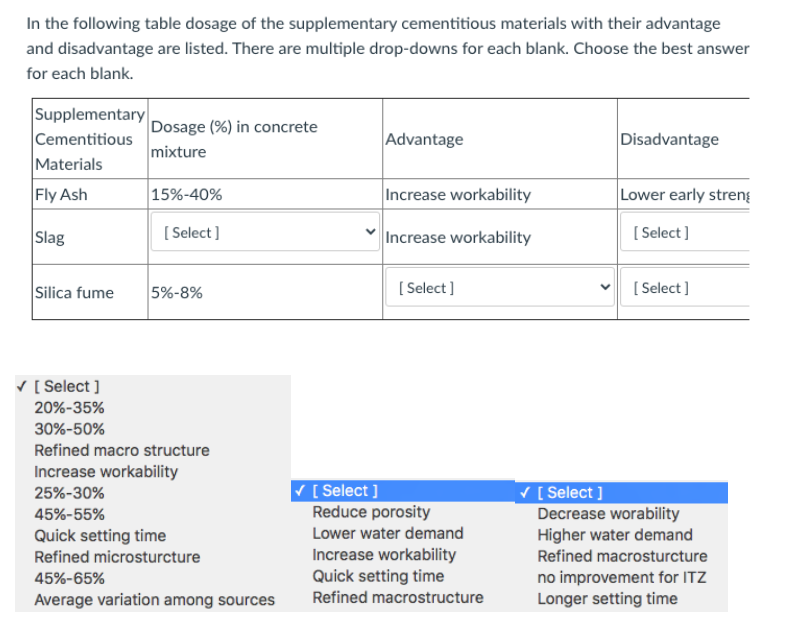
Supplementary Table S1. Normal tissue constraints for treatment plans Structure Constraints Spinal cord maximum dose <45Gy Ki
Supplementary Table 1 Secondary endpoint changes from baseline after 12 wk of treatment (per protocol set) Variable High dose (n
Page 1 SUPPLEMENTARY MATERIALS The Effect of Dose Escalation on the Cost-Effectiveness of Etanercept and Adalimumab with Methotr
Supplementary table 1. Information of dabigatran patient for blood collection Dabigatran patient SE Dabigatran dose Twice a day:
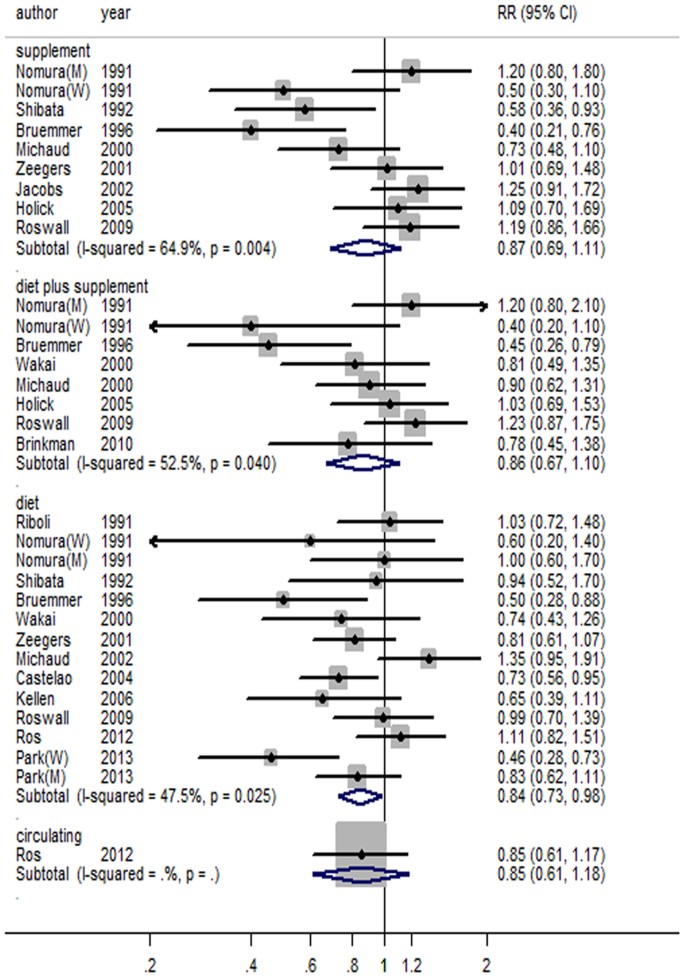
Association of vitamin C, vitamin D, vitamin E and risk of bladder cancer: a dose-response meta-analysis | Scientific Reports
SUPPLEMENTARY MATERIALS Cost-effectiveness of implementing routine hearing screening using a tablet audiometer for pediatric cys
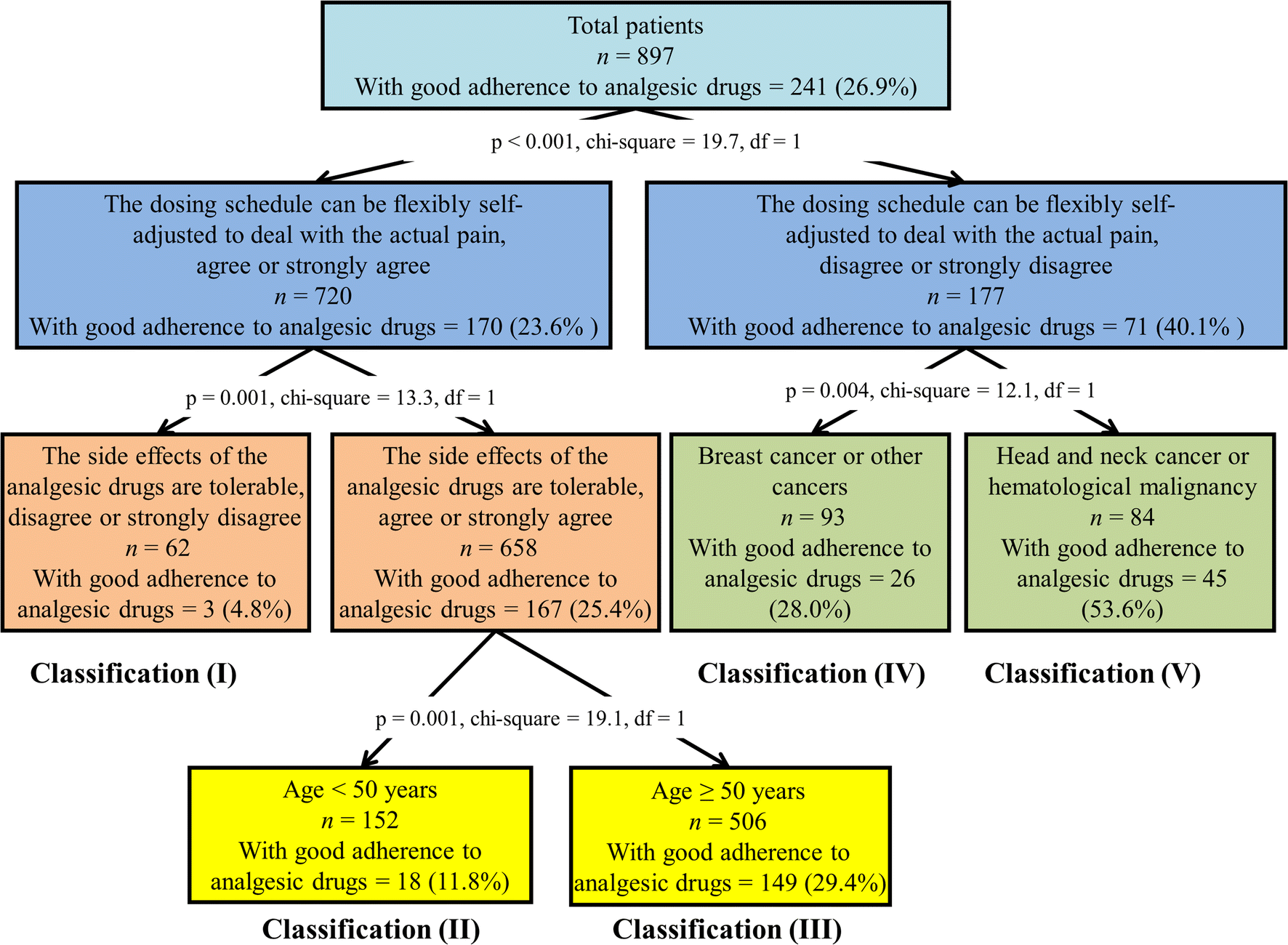
Figure 3 | A nationwide survey of adherence to analgesic drugs among cancer patients in Taiwan: prevalence, determinants, and impact on quality of life | SpringerLink

Clinical Pharmacogenetics Implementation Consortium guideline for CYP2D6 and CYP2C19 genotypes and dosing of tricyclic antidepressants. - Abstract - Europe PMC
Supplementary Table 6. Dose changes during the 1-year observa- tion period Discontinuation group Continuation group Total Dose
Supplementary Table 8. Comparison of Baseline Characteristics between On-Label Standard Dose and Off-Label Underdose Groups in
There is variation within the literature with regards to the recommended dosing in the treatment of vitamin responsive epilepsie